Subblood resuscitation strategy of hemorrhagic shock and traumatic brain injury
![]() 2024-06-11
2024-06-11
Trauma is the leading cause of death in young people. Approximately 40% of civilians in the United States die from traumatic brain injury (TBI), and 33% to 50% of trauma deaths occur before admission. TBI is often associated with massive blood loss and hypotension due to external cranial injury. More than half (about 60%) of all salvage deaths were lost due to hemorrhagic shock (HS).
Even transient hypotension and hypoxemia doubled TBI mortality. The researchers speculated that this was a loss of autoregulatory function, leading to secondary ischemic damage to the already fragile brain. Experimental data also suggest that TBI impairs the physiological compensatory mechanisms of hemorrhagic shock. It is now generally accepted that in patients suffering both TBI and HS, aggressive fluid resuscitation with high-volume crystal solution is necessary with the aim of rapidly dilating blood pressure (BP) and restoring cerebral perfusion pressure (CPP). However, there is considerable evidence that aggressive resuscitation under current general recommendations may lead to failed cerebrovascular hemodynamic optimization, as well as higher short-term mortality. Therefore, the researchers are exploring the use of low-volume surrogate blood (bovine glutaraldehyde cross-linked hemoglobin, HBOC-201 *, formerly Biopure) as a resuscitation solution to treat traumatic brain injury. This drug improves the resuscitation strategy by minimizing the amount of bleeding while maximising tissue oxygenation.
Researchers have conducted extensive research on the use of battlefield and pre-hospital environments. HBOC-201 has been shown to actively resuscitate hemorrhagic shock in the clinic. Several clinical studies have shown that resuscitation fluid based on bovine-derived glutaraldehyde cross-linked hemoglobin (HBOC-201) has a function to dilate vascular content, stabilize hemodynamics through low-volume resuscitation, while delivering oxygen and increase tissue oxygenation, which is similar to that of human red blood cells. However, the interaction between the physiological dysfunction associated with TBI and the prehospital resuscitation of bovine-derived glutaraldehyde-cross-linked hemoglobin has not been further investigated. With multiple organ damage, TBI and hemorrhagic shock often occur simultaneously in the clinic, studying this clause is imperative. Therefore, the investigators designed this study to examine the effect of TBI on surrogate blood resuscitation of hemorrhagic shock in a model of large arterial vascular injury without internal bleeding. The results of this study will help to choose the right resuscitation strategy for patients with multiple trauma.
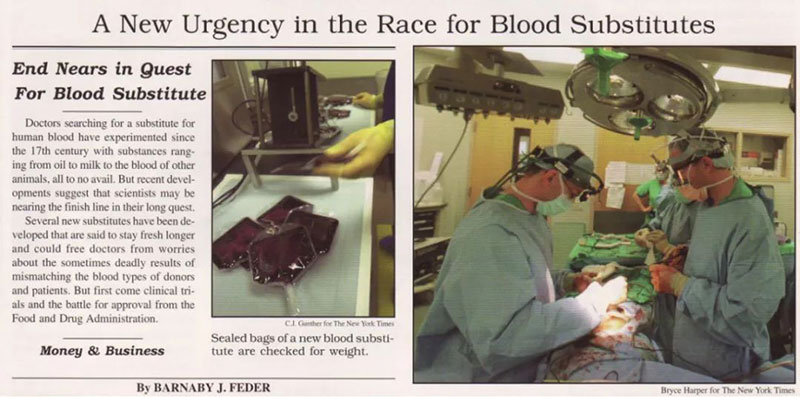
New York Times report on bovine glutaraldehyde crosslinked hemoglobin (photo from Biopure)
【Test methods and results】
These trials were conducted in two phases, each lasting approximately 5 – 6 months. The model used in the first stage was HS + TBI, while the second stage was pure HS. At each session, all animals were randomly assigned to receive one of the two resuscitation solutions (HBOC-201 or lactic ringer).
The present study has two important observations. First, in this multiphasic treatment model, although the presence of TBI was associated with a reduced compensatory response to acute hemorrhage, TBI did not significantly affect the survival of test animals during small-volume resuscitation with noncontrolled hemorrhagic shock. Second, restricted HBOC resuscitation has significantly improved survival rates.
The insufficient effect of TBI on survival may be explained in part by the uneven volume of blood withdrawn during bleeding. Pure hemorrhagic shock animals need to draw an average of 150% more blood to achieve a preset MAP = 30 mmHg. While at the end of the shock phase, similar lactate levels indicated similar levels of shock and an excessive hypotensive response to blood release in the TBI + HS group. Thus, the blood volume of the TBI + HS group animals may apparently remain at higher levels during resuscitation. However, this higher blood volume does not translate into improved MAP, CO, perfusion, or survival time. Taken together, these data suggest that overall vascular reactivity is impaired by TBI during bleeding and resuscitation, counteracting the advantage of increased circulating blood volume. This may also be responsible for the poor prognosis of the TBI + HS animals.
During the resuscitation of hemorrhagic shock, even transient hypotension causes secondary brain injury. Therefore, the excessive hypotensive response to bleeding in patients with TBI may be a clinically relevant phenomenon that deserves further investigation. The study data also suggest that it may be inappropriate to estimate blood loss only based on vital signs if TBI occurs simultaneously. Although vital signs such as heart rate and blood pressure are currently recognized standard methods for determining blood loss and severity of shock in patients with TBI disease, they do not perform well in practice. This study also suggests that estimating blood loss and resuscitation based solely on these parameters alone may lead to ineffective fluid resuscitation.
【conclusion】
In the non-controlled bleeding model, there was no difference in survival between concurrent TBI and HS compared with HS alone. But in the case of TBI, only a smaller amount of bleeding can produce the same level of shock, and the cardiovascular response during fluid resuscitation is correspondingly reduced. These results suggest the existence of a systemic physiology associated with concurrent TBI and hemorrhagic shock that may require a targeted resuscitation strategy. Furthermore, animals had higher survival with limited resuscitation with HBOC compared to lactic ringer's solution.
* Note: HBOC-201 is a chemically stable Bvine colloidal solution polymerized by glutaraldehyde. In 2001, it was licensed in South Africa and Russia, and is the only red blood cell substitute approved in the world. Unlike other products, bovine glutaraldehyde cross-linked hemoglobin can be placed at room temperature (2-30 degrees Celsius) for up to 3 years, with excellent general compatibility and stability.
-

Bios Wins the “Global Hemoglobin Oxygen Carrier Technology Innovation Leadership Award” at the Sullivan New Investment Conference 2025
On August 27, the 19th Sullivan Global Growth, Technology Innovation and Leadership Summit 2025 and the 4th New Investment Conference (hereinafter referred to as “Sullivan New Investment Conference 2025”) was grandly opened in Shangri-La Hotel, Jing ’an District, Shanghai.
2025-08-29
-

Wang Jianfeng, the Secretary of the CPC Changzhou Municipal Committee, met with Mr. Zheng Zhiheng, the Chairman of Bios Biologics
On 28 May 2025, Wang Jianfeng, Party Secretary of Changzhou City, Jiangsu Province, met with Mr. Zheng Zhiheng, Chairman of Bios Biotechnology Co., LTD., Vice Chairman and Executive Director of Chow Tai Fok Jewelry Group Co., LTD., to exchange views on further deepening cooperation.
2025-05-29
-
Yale scientists have successfully revived a pig brain with artificial blood
Recently, the cover of Nature published the latest research from Yale University School of Medicine: scientists at Yale University used an infusion solution to successfully revive the pig brain after 4 hours of death, restoring some cellular and metabolic functions, and maintaining them for at least 6 hours.
2024-08-03
-

A brief history of research and development of red blood cell substitutes
Blood has always been a very important, but very limited medical resource.
2024-08-02


